Unusual presentation of fever and abdominal pain in a 15-year-old female
An undocumented, Spanish-speaking, 15-year-old female presented to the emergency department with 3 weeks of fever (>101℉) and 3 days of abdominal pain. What's the diagnosis?
The case
An undocumented, Spanish-speaking, 15-year-old female presented to the emergency department with 3 weeks of fever (>101℉) and 3 days of abdominal pain. She had previously undergone evaluation at 2 other health care facilities with no resolution of her symptoms. At both encounters, she received a diagnosis of a viral illness and was discharged from the emergency department after fluid administration. On arrival to our facility, vital signs were significant for hypotension, tachycardia, fever of 102.9°F, and tachypnea. The patient was noted to have scars across both knees, scars across the back of her elbows and left arm, and a healing bruise on her lower lip. Initial labs showed microcytic anemia (HgB 9, MCV 79), mild thrombocytopenia (platelets 134,000), elevated ESR (>145), and urinalysis with moderate blood in absence of RBCs. White blood cell count was normal at 6200, as was the absolute neutrophil count at 3000. Blood smear showed normal morphology microcytic anemia, minimally increased absolute lymphocytes without blasts, and mild thrombocytopenia with large platelets. Lactate dehydrogenase, uric acid, amylase, and lipase were normal. Viral respiratory panel was negative. Computed tomography of the abdomen revealed significant intraabdominal and retroperitoneal lymphadenopathy, pericholecystic fluid, and mild splenomegaly.
Significantly, when the patient was aged 4 years, her mother emigrated from Mexico and left her in the care of her teenage sisters and her 40-year-old son-in-law. Patient was brought to the United States 4 years ago because of significant weight loss and poor health. She was currently living in rural Florida with her mother, stepfather, a dog, and a new kitten. Mother was concerned about delinquency and drug use with friends. In the 2 weeks prior to presentation, the patient had been living with a neighbor after an argument with her mother. Patient reports 1 sexual encounter with her boyfriend a few months ago. She admits experimenting with tobacco and marijuana once a few months prior but denies any other drug use.
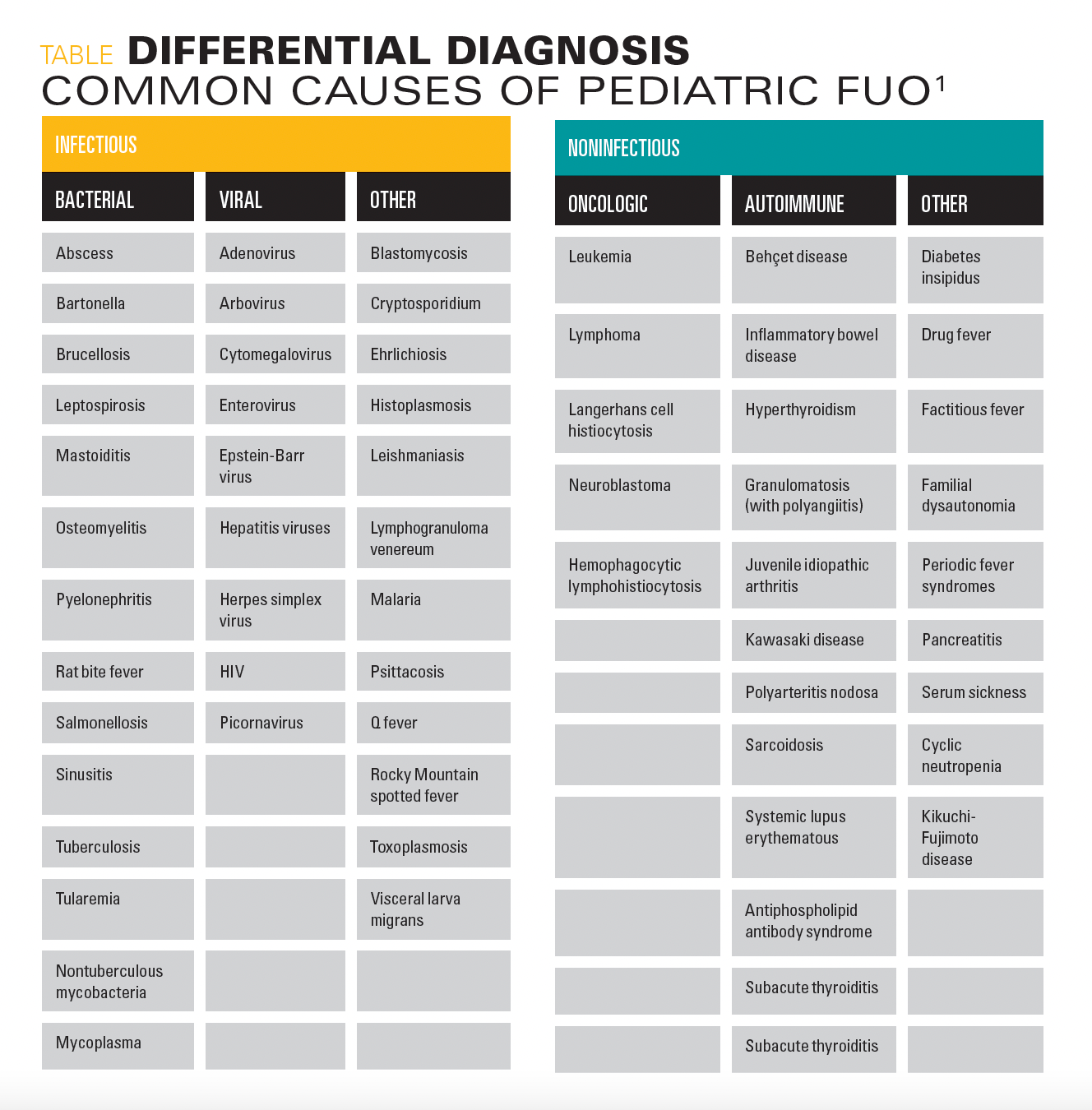
Differential diagnosis
Fever of unknown origin (FUO) has been defined as a temperature higher than 38.0 °C (100.4 °F) that lasts at least 8 days without a clear source.1 The 3 most common etiologic categories of FUO in children in order of frequency are infectious diseases, connective tissue diseases, and neoplasms.1Outlined in the differential diagnosis table are etiologies of FUO by category.1 Infectious diseases to consider in immigrant children include Mycobacterium tuberculosis, Mycobacterium bovis, hepatitis A-D, parasitic infections, malaria, and typhoid fever.2,3
Hospital course/diagnosis
Patient was stabilized with maintenance intravenous fluids within 24 hours. She was given acetaminophen and ibuprofen as needed with adequate control of her pain. The workup of FUO was completed to guide further management.
Patient’s tests returned positive for Bartonella henselae IgM and dsDNA IgG with significant ANA titer of 1:640. HIV antigen and antibody testing returned positive with CD4 count of 200, CD4 percentage of 4%-11%, and quantitative HIV RNA count of 272,000/ml. Subsequent testing for other sexually transmitted infections returned positive for syphilis with reactive rapid plasma reagin (RPR), but RPR titers were negative, conflicting for true syphilis infection. Treponema pallidum particle agglutination assay (TP-PA) was performed as confirmatory testing and the positive result suggested either very early disease with no measurable seroconversion, late or late latent syphilis, or previously treated syphilis. She had no symptoms suggestive of active disease, and there was no report or documentation of treatment. This led to the diagnosis of HIV with latent syphilis infection and immune dysregulation resulting in multiple false-positive antibody titers.
Discussion
This patient’s FUO workup led to several positive tests that could account for her fevers, including Bartonella, systemic lupus erythematosus (SLE), HIV, and syphilis. We were faced with a diagnostic and treatment dilemma—to treat or not to treat? Of the plausible diagnoses, acute HIV was most likely based on symptomatology. Acute retroviral syndrome develops in 50% to 90% of adolescents and adults within the first few weeks of HIV infection and can mimic mononucleosis because of fever, malaise, lymphadenopathy, and skin rash.4 Untreated pediatric HIV infection can present as unexplained fevers, generalized lymphadenopathy, hepatomegaly, splenomegaly, and failure to thrive4, findings similar to those in our patient.
Classic symptoms of Bartonella include a papule or pustule at site of inoculation within 12 days of a cat scratch, followed by regional lymphadenopathy within 1 to 2 weeks in nodes that drain site of inoculation. Axillary node involvement is most common, but cervical, submandibular, submental, epitrochlear, or inguinal nodes can be involved and can last up to 4 months. Low-grade fever lasting several days develops in 30% of patients.5 The updated diagnostic criteria for SLE includes a positive ANA titer of ≥ 1:80 or an equivalent positive test, presence of 1 clinical criterion, and ≥ 10 points on scoring system defined by symptoms and/or diagnostic testing. Clinical domains for diagnosis include constitutional, hematologic, neuropsychiatric, mucocutaneous, serosal, musculoskeletal, and renal symptoms. Antiphospholipid antibodies, complement proteins, and SLE-specific antibodies (anti-dsDNA antibody or anti-Smith antibody) are also included in diagnostic criteria.6 The absence of clinical findings for Bartonella and lack of diagnostic criteria for SLE prompted the decision to treat HIV and syphilis first. Ultimately, significant Bartonella, dsDNA, and ANA titers were thought to be a result of immune dysregulation.
Immune dysregulation in the setting of HIV infection is an interesting phenomenon. HIV viremia is known to result in B cell hyperactivity, which results in hypergammaglobulinemia of both IgG and IgM antibodies7,8 and increased production of autoantibodies. This can result in false positive antibody testing for various diseases. Antibody cross reactivity with tuberculosis, leprosy, and malaria in the setting of HIV has been reported.9 HIV has also been associated with autoimmune disease, especially following initiation of antiretroviral therapy and immune system recovery.10 However, a subset of these patients have demonstrated antibody positivity in THE absence of clinical symptoms suggestive of disease.10The exact mechanism is unclear. There are a few proposed mechanisms of B cell activation including (1) direct or indirect activation of B cells by cytokines and growth factors in HIV-infected patients or (2) activation by HIV proteins or the virus itself. False positive testing for autoantibodies, as detected by indirect immunofluorescence techniques, may reflect nonspecific IgG binding to Fc receptors.8
Within 12 weeks of starting antiretroviral therapy for HIV, subclinical opportunistic infections can be unmasked as immune function improves.11 Follow-up and surveillance of opportunistic infections are critical. Bartonella is a known opportunistic infection with complications including seronegative endocarditis, bacillary angiomatosis (vascular proliferative lesions of skin and subcutaneous tissue), and bacillary peliosis (reticuloendothelial lesions in visceral organs, primarily the liver)4, so development of symptoms suggestive of bartonella infection would have warranted treatment. Other bacterial opportunistic infections including invasive Streptococcus pneumoniae, Haemophilus influenzae type B, and meningococcal infections would also warrant treatment if clinically appropriate.12
Latent syphilis, observed in this patient, is defined as the period after infection when the patient is seroreactive but demonstrates no clinical manifestations of disease. Latent syphilis acquired within the preceding year is referred to as early latent syphilis; cases greater than 1 year’s duration are considered late latent syphilis. Traditional serologic testing for syphilis involves initial screening with a nontreponemal test like RPR, followed by confirmatory testing for a positive test with a treponemal test, such as TP-PA.13 An increasingly popular algorithm reverses the order and uses a treponemal antibody test as the screening test followed by a nontreponemal test (eg, RPR) for confirmation.14 This algorithm has gained popularity because it has been shown to detect syphilis in some patients who would not have been identified if a nontreponemal test were used initially. If an initial treponemal antibody test is positive, an RPR can be performed to differentiate between an active or past infection. In this case, a positive RPR would confirm that the person has been exposed to syphilis and, if not treated previously, has an active infection; or, if treatment had occurred more than 3 years ago, possible reinfection.Discordant results (eg, positive antibody test, negative RPR) are often seen in patients with a history of successfully treated syphilis. For patients without a history of treated syphilis, a discordant result can occur in very early syphilis or in late syphilis when nontreponemal tests have become nonreactive over time.14 This patient had a reactive RPR with negative RPR titers but had a positive TP-PA, and thus was diagnosed with latent syphilis under the presumption she was not treated and the nontreponemal test had become nonreactive over time. It is important to treat syphilis, even in latent syphilis, because a subset of patients will develop tertiary syphilis if untreated.15 Tertiary syphilis occurs 15 to 30 years after the initial infection and can include gumma formation (soft, noncancerous, granulomatous growths that can destroy tissue) or cardiovascular involvement (including aortitis).13 It can also damage the brain, nerves, eyes, blood vessels, liver, bones, and joints.16 Risk of neurosyphilis is high in HIV patients and can occur at any stage.13
Given this patient’s social situation, the health care team had significant concerns about the patient’s safety and risk for human and sex trafficking especially in the setting of HIV diagnosis and starting antiretroviral therapy. Red flags included the mother not resembling the child and the patient revealing that she could speak English several days into admission. Major risks for human trafficking include age between 12 and 16 years, homelessness, substance use, history of sexual abuse, LGBT identification, and mental illness.17 Her undocumented status—a social determinant of health—puts her at risk for unsafe work and living conditions, migration-related trauma, violent injury, sexual assault, and barriers/delay to accessing health care because of fear of detention and deportation.18 Careful attention was given to discharge planning. Safe discharge criteria for this patient included addressing social and medical issues. The state Department of Children and Families (DCF) was contacted and a mandatory report was made for concern of patient’s safety. Although HIV positivity could be consistent with sexual contact reported by the patient several months ago, latent syphilis diagnosis suggests acquisition time inconsistent with history. A safety visit was made to the patient’s home followed by clearance from social work and DCF with development of a safe discharge and follow-up plan.
Patient outcome
The patient was started on antiretroviral therapy with daily bictegravir (Biktavry) for HIV and penicillin G benzocaine IM once a week for 3 weeks for latent syphilis, and was monitored for signs and symptoms of Bartonella and SLE. Daily Bactrim was started because of the risk for opportunistic Pneumocystis jiroveci pneumonia infection for CD4 count <200. The patient was accepted into the Health Resources and Services Administration’s Ryan White HIV/AIDS Program, which provides a comprehensive system of HIV primary medical care, essential support services, and medications for low-income people living with HIV who are uninsured and underserved. Her long-term care and follow-up were arranged at her local health department and provided by the AIDS Drug Assistance Program (ADAP), a statewide, federally funded prescription medication program for uninsured or underinsured individuals living with HIV. Treatment of HIV and syphilis alone resulted in complete symptomatic improvement.
BIO
Autumn Hinds is a pediatric resident at Nemours Children’s Hospital in Orlando, Florida.
Acknowledgements
The author thanks Usa Gift Kopsombut, MD, a pediatric hospitalist at Nemours Children’s Hospital, for her expertise, review and editing of the manuscript.
References:
1. Antoon JW, Potisek NM, Lohr JA. Pediatric fever of unknown origin. Pediatr Rev. 2015;36(9):380-391. doi:10.1542/pir.36-9-380
2. Council on Community Pediatrics. Providing care for immigrant, migrant, and border children. Pediatrics. 2013;131(6):2028-2034. doi:10.1542/peds.2013-1099
3. Shetty AK. Infectious diseases among refugee children. Children. 2019;6(12):129. doi:10.3390/children6120129
4. Kimberlin DW, Barnett E, Lynfield R, Sawyer MH. Section 3: Summaries of infectious diseases—human immunodeficiency virus infection. In:Red Book 2021. 32nd ed. American Academy of Pediatrics. 2021:427-440.
5. Kimberlin D, Barnett E, Lynfield R, Sawyer M. Section 3: Summaries of infectious diseases—Bartonella henselae (cat-scratch disease). In: Red Book 2021. 32nd ed. American Academy of Pediatrics; 2021:226-229.
6. Aringer M, Costenbader K, Daikh D, et al. 2019 European League Against Rheumatism/American College of Rheumatology classification criteria for systemic lupus erythematosus. Arthritis Rheumatol. 2019;71(9):1400-1412. doi:10.1002/art.40930
7. Davenport A, Grant PJ. False positive auto-antibodies in HIV infection. Nephron. 1991;59(3):515-516. doi:10.1159/000186626
8. Moir S, Fauci AS. B-cell responses to HIV infection. Immunol Rev. 2017;275(1):33-48. doi:10.1111/imr.12502
9. Swaminathan S, Hanna LE, Sundaramurthi JC, et al. Prevalence and pattern of cross-reacting antibodies to HIV in patients with tuberculosis.AIDS Res Hum Retroviruses. 2008;24(7):941-946. doi:10.1089/aid.2007.0211
10. Zandman-Goddard G, Shoenfeld Y. HIV and autoimmunity. Autoimmun Rev. 2002;1(6):329-337. doi:10.1016/s1568-9972(02)00086-1
11. Moir S, Fauci A. B cells in HIV infection and disease. Nat Rev Immunol. 2009;9(4):235-245. doi:10.1038/nri2524
12. Guidelines for the prevention and treatment of opportunistic infections among HIV-exposed and HIV-infected children: recommendations from CDC, the National Institutes of Health, the HIV Medicine Association of the Infectious Diseases Society of America, the Pediatric Infectious Diseases Society, and the American Academy of Pediatrics. MMWR Recomm Rep. 2009;58(RR-11):1-166. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2821196/
13. Kimberlin D, Barnett E, Lynfield R, Sawyer M. Section 3: Summaries of infectious diseases—syphilis. In: Red Book 2021. 32nd ed. American Academy of Pediatrics; 2021:729-744.
14. Peterman T, Schillinger J, Blank S, et al. Syphilis testing algorithms using treponemal tests for initial screening—four laboratories, New York City, 2005-2006. MMWR Weekly. Centers for Disease Control and Prevention. 2008;57(32):872-875. August 15, 2008. Accessed May 2, 2022. https://www.cdc.gov/mmwr/preview/mmwrhtml/mm5732a2.htm
15. Syphilis — CDC fact sheet (detailed). Centers for Disease Control and Prevention. April 22, 2021. Accessed August 17, 2021. https://www.cdc.gov/std/syphilis/stdfact-syphilis-detailed.htm
16. Syphilis. Mayo Clinic. Accessed August 20, 2021. https://www.mayoclinic.org/diseases-conditions/syphilis/symptoms-causes/syc-20351756?p=1
17. Greenbaum J, Crawford-Jakubiak JE; Committee on Child Abuse and Neglect. Child sex trafficking and commercial sexual exploitation: health care needs of victims. Pediatrics. 2015;13(3):566-574. doi:10.1542/peds.2014-4138
18. Samra S, Taira B, Pinheiro E, Trotzky-Sirr R, Schneberk T. Undocumented patients in the emergency department: challenges and opportunities. West J Emerg Med. 2019;20(5):791-798. doi:10.5811/westjem.2019.7.41489
Newsletter
Access practical, evidence-based guidance to support better care for our youngest patients. Join our email list for the latest clinical updates.
Having "the talk" with teen patients
June 17th 2022A visit with a pediatric clinician is an ideal time to ensure that a teenager knows the correct information, has the opportunity to make certain contraceptive choices, and instill the knowledge that the pediatric office is a safe place to come for help.
Meet the Board: Vivian P. Hernandez-Trujillo, MD, FAAP, FAAAAI, FACAAI
May 20th 2022Contemporary Pediatrics sat down with one of our newest editorial advisory board members: Vivian P. Hernandez-Trujillo, MD, FAAP, FAAAAI, FACAAI to discuss what led to her career in medicine and what she thinks the future holds for pediatrics.