Newborn with midline neck lesion
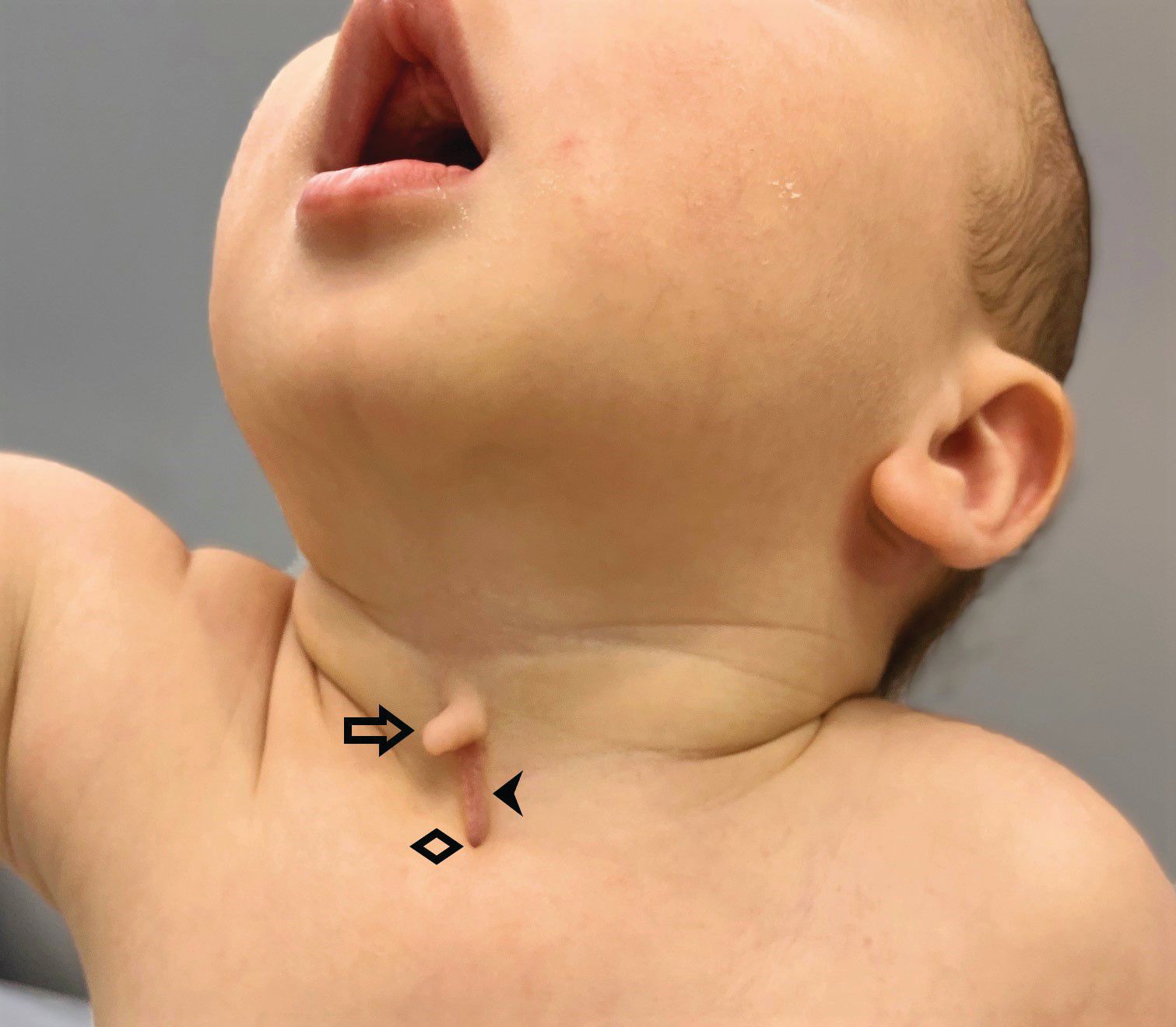
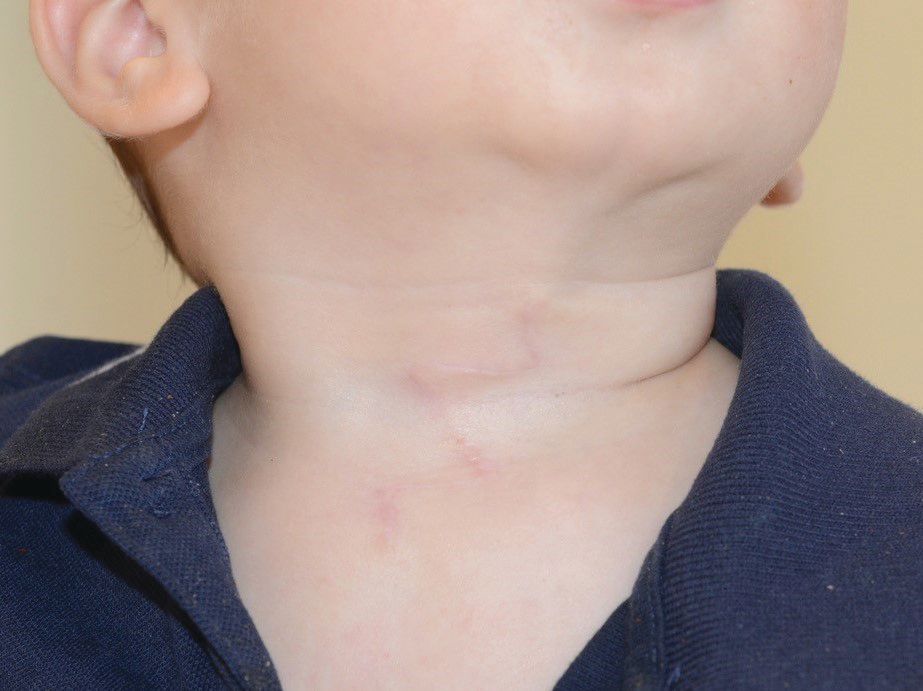
A 4-day-old boy with a midline neck lesion was born at term by normal vaginal delivery. After birth, mid line lesion had the configuration of a linear cleft with a cephalocaudal orientation, extending from the level below the hyoid bone to the suprasternal notch with a length of 2.5 cm and a width of 0.5 cm. What's the diagnosis?
(Figure 1)
Newborn with midline neck lesion | Image Credit: Author provided
Presentation
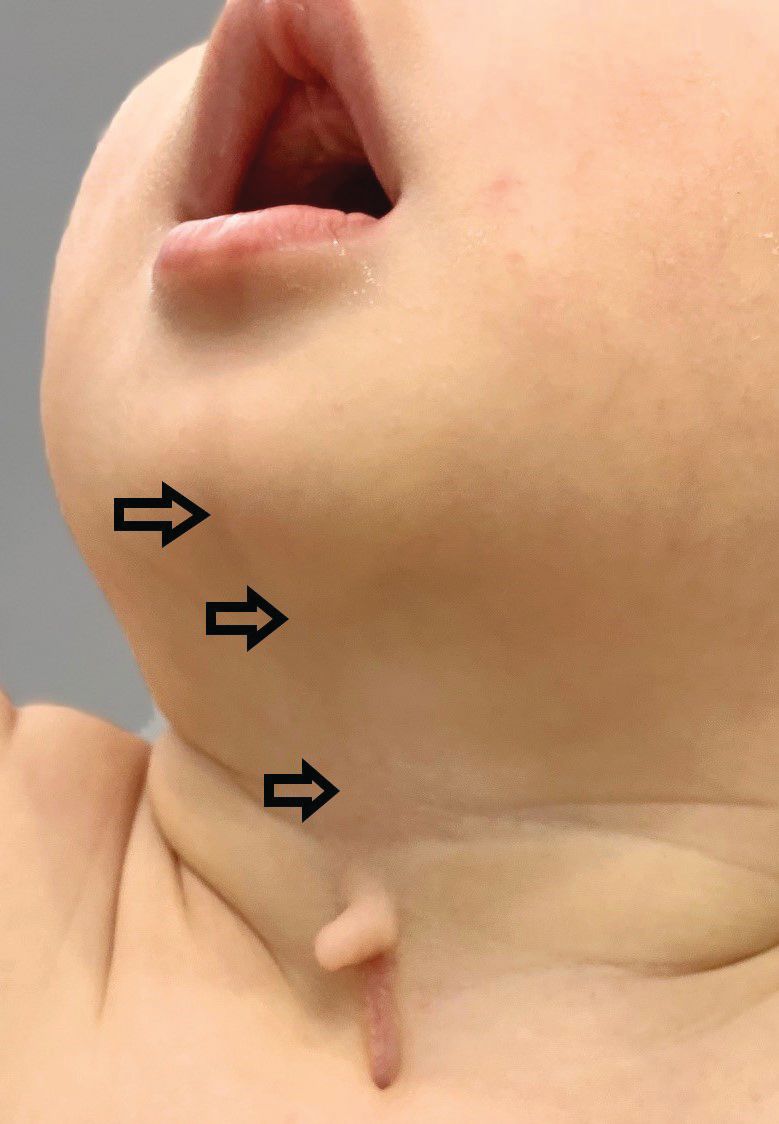
A 4-day-old boy with a midline neck lesion (Figure 1) was born at term by normal vaginal delivery. Antenatal fetal scans were normal except for the midline neck lesion. After birth, mid line lesion had the configuration of a linear cleft with a cephalocaudal orientation, extending from the level below the hyoid bone to the suprasternal notch with a length of 2.5 cm and a width of 0.5 cm (Figure 1). It was composed of 3 components: a nipplelike projection (Figure 1, black arrow), midline longitudinal sulcus (Figure 1, solid black arrowhead) and a blind sinus (Figure 1, black diamond) at the inferior end. There was a seromucinous discharge from the sinus, which was gradually reduced during few weeks of infancy. On extension of the neck, a skin web was formed between the skin lesion and the mandible (Figure 2).
(Figure 2)
Neck extension produces webbing of the skin between the cleft and the mandible (black arrow).
He was clinically well with a normal medical examination and no evidence of any feeding or breathing issues, or noisy respirations. There was no family history of similar lesions.
Diagnosis: congenital midline cervical cleft
Congenital midline cervical cleft (CMCC) is a rare developmental defect of the anterior neck region over the midline and accounts for 2% of congenital cervical malformations. It was first reported in 1848 by Luschka and described by Bailey in 1924.1 It’s mostly seen in Caucasian females with a female to male ratio of 2:1. CMCC is not considered a true cleft since no skin gap exists. It represents a variant of the cleft category number 30 of the Tessier classification system of craniofacial defects. The cardinal diagnostic features (Figure 1) are as follows:
(a) fistula opening is located caudally;
(b) intermittent serous fluid discharge in the early neonatal period;
(c) nipple-like appearance of the cleft in the superior aspect;
(d) thin vertical band of erythematous skin
(e) subcutaneous cord-like fibrous thickening cephalad to the defect (Figure 2), which may cause webbing, also known as pterygium colli mediastinum.2
In addition to its midline presentation, CMCC may be associated with other anomalies of the head and neck including clefts of the lower lip, mandible, chin and tongue, and hypoplasia/aplasia of the hyoid bone. It may be associated with cleft sternum, midline abdominal web, and infantile hemangioma as well as congenital heart defects such as ectopia and intracardiac anomalies.

Embryology and etiology
Branchial apparatus development begins by the fourth week of gestation and is completed by week 6. It consists of 6 pairs of mesodermal arches lying in the transverse plane of the neck, numbered from the cranial to caudal direction. The fifth arch is small and regresses early. The rest of the arches are separated internally by endoderm-lined pouches and externally by ectoderm-lined clefts. Normally they merge medially from the cephalad to caudal direction. The exact pathogenesis of congenital midline cervical clefts remains speculative. The most widely accepted theory is impaired fusion of the first, or, most commonly, second branchial arches in the midline as well as improper interaction between the ectoderm and mesoderm.3 In CMCC, some mechanical or vascular anomaly is thought to result in ischemia or necrosis and interfere with these normal processes. Other less accepted explanations include increased pressure in the cervical region from the developing pericardial roof, amniotic adhesions, or exteriorization of a thyroglossal duct cyst.4 There are no other etiological risk factors. Improper interaction between the mesoderm and ectoderm could explain the absence of skin adnexal structures.
Clinical features
CMCC presents at birth as a ventral midline defect of the skin of the neck. It appears as a thin vertical band of erythematous skin at any level between the chin and sternal notch, with a nipplelike projection superiorly. There will be an associated sinus tract at the caudal aspect, which may discharge mucoid material. There always exists a subcutaneous cordlike, fibrous thickening cephalad to the defect, which may lead to a neck contracture and eventually torticollis or micrognathia in early childhood.5
Diagnosis and investigations
CMCC is usually diagnosed by characteristic clinical features as described above. Additional imaging studies are mostly useful for confirmation and differencing from similar skin lesions nearby (Table) such as thyroglossal cyst, branchial cyst/fistula, dermoid cyst, and others. Neck ultrasound shows a nonvascular blind-ending sinus tract from the skin surface, extending caudally. The thyroid gland is normal. MRI usually demonstrates skin thinning and a peripherally enhancing T1 hypointense and T2 hyperintense linear tract without disruption of any bony or cartilaginous structures or involvement of the thyroid gland.6 Histologic examination shows stratified keratinized squamous epithelium lacking skin appendages such as sweat or sebaceous glands. The dermis has alternating hypertrophy and atrophy, and there is a mild lymphocytic inflammatory infiltrate in the subcutaneous tissues.7
8Thyroglossal duct cysts are the most common form of congenital cysts in the neck. These cysts are epithelial remnants of the thyroglossal tract and characteristically appears as midline cysts at the level of thyrohyoid membrane, which are closely attached to hyoid bone. They often present as a soft, mobile, painless cystic mass that moves with swallowing and protuberance of the tongue. They usually present as a mass lesion or focus of infection with pain, sore throat, dysphagia, hoarseness, globus, and fistula formation.8 Ultrasound of neck reveals a well-defined, thin walled, anechoic or hypoechoic mass with posterior enhancement. On MRI, uncomplicated thyroglossal duct cyst appears dark on T1-weighted images and bright on T2-weighted images, reflecting the fluid content of the cyst. In patients with ectopic thyroid tissue, radionucleotide imaging (thyroid scan) or CT/MRI are needed to identify the presence of normal thyroid gland and help to rule out ectopic thyroid tissue. All thyroglossal duct cysts should be treated with the sistrunk procedure.
Branchial cleft cyst anomalies usually arise from the first and second branchial arches. They are usually located anterior to the sternocleidomastoid muscle on the lower third of the neck. The sinus tract of the second branchial cleft cyst will travel through the deep structures of the neck and open into the tonsillar fossa. Ultrasound imaging shows anechoic or hypoechoic cyst posterolateral to submandibular gland and anterior to sternocleidomastoid muscle. Recurrent infections of branchial cysts can occur and complicates with a fistula track with repeated infections. Management of branchial cleft cyst include control of infection followed by surgical excision of the cyst.
Dermoid cysts are congenital lesions that present as subcutaneous nodules seen along the embryonic fusion lines on the face, spine, scalp, and midline neck, and contains epidermal and dermal tissues. Overlying skin will be normal or may contain pit/sinus. They are usually adherent to the periosteum and do not move with swallowing or protrusion of the tongue. Patients with midline lesions should be evaluated with neuroimaging to detect central nervous system connections or underlying spinal dysraphism. Dermoid cysts should be surgically excised because of the risk of associated complications.
Treatment
CMCC is well treated with surgery for aesthetic purposes and to prevent development of cervical contractures, mandibular hypoplasia, and other defects. Surgical procedures consist of the excision of skin, fibrous card, and caudal sinus, and removal of the cyst. Methods of closure are linear closure, W-plasty, single Z-plasty, double Z-plasty, and opposing Z-plasty.9 A simple linear vertical closure can be done if the defect is small, but it might lead to contracture formation and limitations in neck movement. Z-plasty is a common technique and allows the surgeon to lengthen the contracted scar, reorient the direction of a scar or defect, break up a straight line, and shift soft tissue contour.10 The Z-plasty is symmetrically designed so that the lateral limbs are equal in length to the tissue defect (central limb) and the angles between the lateral limbs and the central limb are 60 degrees. The angle may be greater than 60 degrees, but the tension required to transpose skin flaps increases as the angle of the Z-plasty increases, such that angles greater than 75 degrees cause tissue distortion and dog-ear deformities. Angles less than 20 degrees present problems with flap viability secondary to compromised blood flow at the flap tips.11 Z-plasty closure improves functional results and has a positive effect on neck contracture. Recurrences are uncommon and CMCC lesions are generally benign.
(Figure 3)
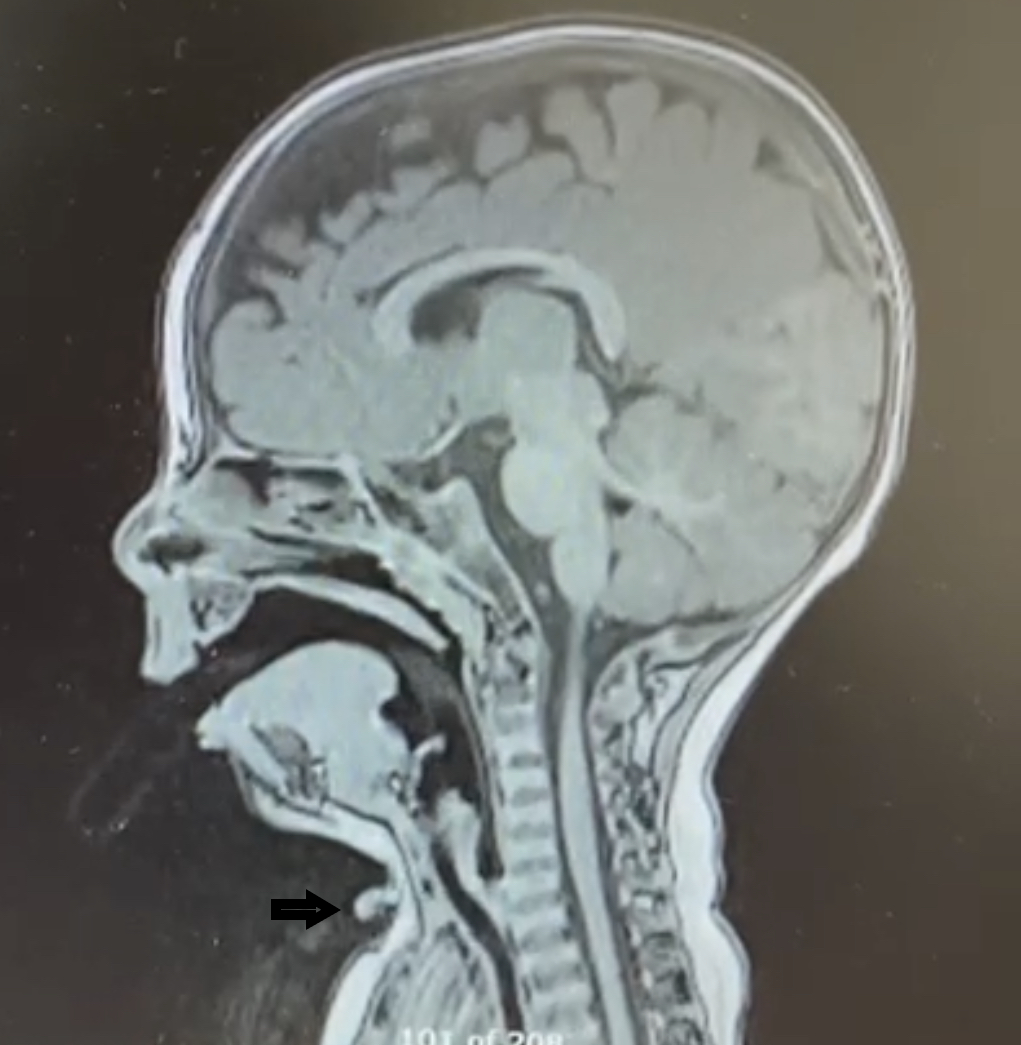
Sagittal T2 -weighted MRI image of the patient showing midline cervical cleft on vental side (Black arrow).
(Figure 4)
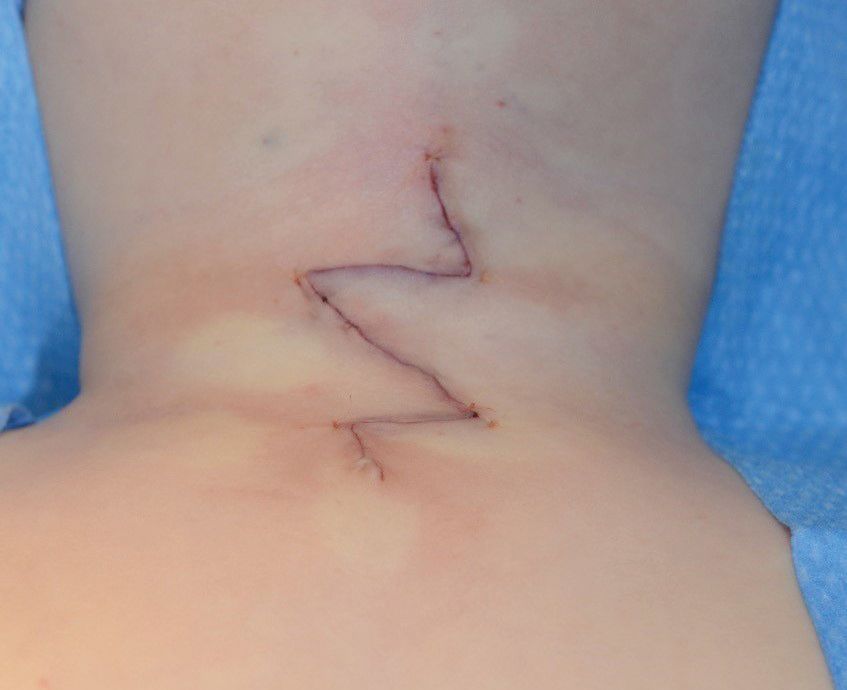
Z plasty postoperative wound closure.
(Figure 5)
Wound exam at follow up. Note the full extension of neck and nice healing of the Z-plasty wound.
Clinical Follow-Up
The patient was evaluated at the age of 2 months by a plastic surgeon and surgical excision was planned for between the ages of 6 months and 12 months. Preoperative pre- and postcontrast MRI of the neck (Figure 3) showed a midline anterior cervical cleft at the C7 level, with a defect in the epidermis associated with a superior skin tag measuring approximately 7 mm in length, extending superiorly from the cleft with a thin midline cord of mixed T1 signal appearance of fibrous and fatty components reaching the submental subcutaneous tissues. Separate from this, extending inferiorly from the cleft was a linear T1 hypointense signal terminating in a T1 hypointense, T2 hyperintense cystic-appearing structure within the subcutaneous tissues at the midline and suprasternal notch (Figure 2) measuring 5.4 mm. The lesion overlaid the superior aspect of the manubrium without evidence of a thyroglossal duct anomaly. A linear structure extending superiorly from the cleft, reaching the submental cutaneous tissues, represented a fibrous cord, a known association of midline cervical cleft. Surgical excision was performed at the age of 9 months. The entire lesion, including the caudal sinus and underlying cephalocaudal fibers, was excised and sutured with a Z-plasty (Figure 4). At follow-up a few months later, he had a well-healed cosmetic scar (Figure 5) without any contracture of the adjacent skin or limitation of neck extension.
Conclusions
Various congenital malformations can affect the anterior part of the neck at birth and often present as a diagnostic challenge for the pediatrician, primary care physician, pediatric surgeon, and otolaryngologist. CMCC represents a rare defect of the midline neck, which is sometimes wrongly misdiagnosed as a thyroglossal duct anomaly, dermoid cyst, branchial cleft anomaly, or miscellaneous birthmark. Prompt clinical diagnosis and early surgical intervention are essential to ensure a good functional and esthetic outcome and prevent long-term complications of neck movement restrictions, cervical tethering, or mandibular defects. A complete excision with a tension-free skin closure yields optimal results.
For more from the November/December issue of Contemporary Pediatrics, click here.
References
1. Jaiswal AA, Behera BK, Membally R, Mohanty MK. Congenital midline cervical cleft: a case
report with review of literature. Int J Head Neck Surg. 2017;8(1):25-30. doi:10.5005/jp-journals-10001-1301
2. Saha S, Misra S, Saha VP, Mondal AR. Midline cervical cleft: a report of two cases. Indian J
Otolaryngol Head Neck Surg. 2005;57(1):78-79. doi:10.1007/BF02907640
3. Van der Staak F, Pruszczynski M, Severijnen R, Van de Kaa CA, Festen C. The midline cervical cleft. J Pediatr Surg.1991;26(12):1391-1393. doi:10.1016/0022-3468(91)91042-w
4. Mlynarek A, Hagr A, Tewfik TL, Nguyen VH. Congenital mid-line cervical cleft: case report and review of literature. Int J Pediatr Otorhinolaryngol. 2003;67(11):1243-1249. doi:10.1016/s0165-5876(03)00201-5
5. King J, Patel RV, Huddart SN. Congenital midline cervical cleft. J Pediatr Surg Case Reports.
2013;1(5):99-101. doi:10.1016/j.epsc.2013.04.002
6. Villanueva-Meyer J, Glastonbury C, Marcovici P. Congenital midline cervical cleft. J Radiol Case Rep. 2015;9(3):7-11. doi:10.3941/jrcr.v9i3.2202
7. Celikoyar M,Aktan E, Dogusoy G. Congenital midline cervical cleft: a case report.
J Med Case Rep.2019,Jun 9;13(1):176. Doi :10.1186/s 13256-019-2116-6. PMID:31176376;PMCID:PMC6556223.
8. Crippa BL, Bedeschi MF, Cantarella G, et al. Congenital midline cervical cleft: clinical approach to a congenital anterior neck defect. Congenit Anom (Kyoto). 2015;55(2):112-115. doi:10.1111/cga.12086
9. Daw JL Jr, Patel PK. Double–opposing Z-plasty for correction of midline cervical web. J Craniofac Surg. 2003;14(5):774-778. doi:10.1097/00001665-200309000-00032
10. Hudson DA. Some thoughts on choosing a Z- plasty: the Z made simple. Plast Reconstr Surg. 2000;106(3):665-671. doi:10.1097/00006534-200009030-00024
11. Davis WE, Schrick RE, Templer J. An introduction to Z-plasty. Ear Nose Throat J. 1981;60(1):29-34.

Newsletter
Access practical, evidence-based guidance to support better care for our youngest patients. Join our email list for the latest clinical updates.
Full case: Infant presents with an asymptomatic pearl-like nodule on the heel
Infant is closely monitored at subsequent well visits and rechecked after 3 months, showed full resolution of the skin lesion over the heel area.
Diagnose this infant with an asymptomatic pearl-like nodule on the heel
Can you diagnose this patient? Take our poll and find out! Then check back for the full case, differential diagnosis, and correct diagnosis.