Vaccination and Immunization
Latest News
Latest Videos

Shorts
Podcasts
CME Content
More News

“We started out 2025 with an environment that was rife with myths and disinformation," said Robert H. Hopkins, Jr., MD, in this year-end recap of 2025 and the pediatric vaccine landscape.

Donna Hallas, PhD, PPCNP-BC, CPNP, PMHS, FAANP, FAAN, shares her thoughts on the recently updated ACIP recommendation on the hepatitis B vaccine.

According to multiple reports, the FDA is investigating the safety of a pair of already-approved protective treatments for respiratory syncytial virus.
“We took a different approach by looking at colonizing bacteria, and we found vaccination reduced antimicrobial resistance through a completely different mechanism," said Brooke Ramay, lead author of the study.

The ACIP committee voted to pass both resolutions to update its recommendation on the hepatitis B vaccine for infants.

The CDC website now claims "studies supporting a link [between vaccines and autism] have been ignored by health authorities."
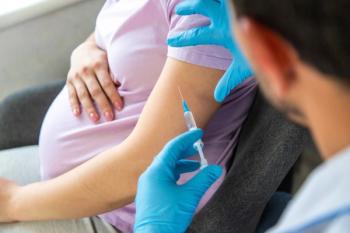
COVID-19 vaccination in pregnancy reduced infection, stillbirth, and preterm birth risks without increased adverse maternal outcomes.

Plus: The new Contemporary Pediatrics interactive journal…have you experienced it?

Members of the Contemporary Pediatrics Editorial Advisory Board react to the Advisory Committee on Immunization Practices' September 2025 meeting.

The AAP recommends that all children aged 6 months to 23 months receive a COVID-19 vaccine, as this group has the highest risk for severe infection.

As families prepare for a new school year, pediatricians are essential in helping parents navigate immunization schedules and school requirements and ensuring vaccine confidence.
New CDC data show fewer kindergartners are fully vaccinated as exemption rates increased during the 2024-2025 school year.
Pediatricians are urged to follow 2024 guidelines amid ACIP upheaval, vaccine hesitancy, and access issues as children head back to school.

Get caught up with Contemporary Pediatrics! This list helps you navigate our top stories from the week, all in one place.

This week, HHS Secretary Robert F. Kennedy Jr signed the ACIP's recommendation to remove the mercury-based preservative from all influenza vaccines.
A newly-published study identified specific groups of higher-risk children that could benefit most from monoclonal antibodies.

The final recommendation sign off decision will go to HHS secretary Robert F. Kennedy Jr.
The American Academy of Pediatrics will continue to hold its own childhood vaccine schedule, as it has since the 1930s.

Donna Hallas, PhD, PPCNP-BC, CPNP, PMHS, FAANP, FAAN, shares her thoughts on how PNPs can implement evidence-based strategies to encourage parents to vaccinate their children.

Currently, Vimkunya is FDA-approved for use among individuals aged 12 years and older.

Ramilo, a clesrovimab clinical trial investigator, breaks down the recent FDA approval for the newest monoclonal antibody to protect against RSV disease in infants.

The BLA for clesrovimab was based on results demonstrated in the phase 2b/3 CLEVER trial among healthy pre-term and full-term infants.
In this article, we recap a timeline of recent federal agency changes to routine COVID-19 vaccination intended for the pediatric population.

Federal health agencies have adjusted COVID vaccine guidance, potentially changing eligibility and access for pediatric patients. Experts respond in this article.
New phase 3b durability data revealed nirsevimab reduced RSV hospitalizations in infants by 82.7% through 180 days vs no intervention.