
The landmark indication is supported by a couple of pivotal trials assessing dupilumab for patients with chronic rhinosinusitis with nasal polyps, as well as a pediatric severe asthma trial assessing the biologic.

The landmark indication is supported by a couple of pivotal trials assessing dupilumab for patients with chronic rhinosinusitis with nasal polyps, as well as a pediatric severe asthma trial assessing the biologic.

The indication is supported by trial data showing three-fourths of successfully treated patients maintained clear skin through 1 year of a lebrikizumab regimen.

A new study suggests an early AI-powered algorithm could help accurately identify common movements exhibited by children with autism spectrum disorder.

The CDC committee has set new recommendations based on children's age, vaccination status, and overall immunity health, for the 2024 - 2025 period.

A cohort analysis shows state Medicaid programs could assure young children with sickle cell disease receive cost-efficient and critical care in a timely fashion.

A laboratory-based assessment shows children aged 18 - 32 months old were less likely to listen to attention prompts or their parent's requests when provided an iPad versus a standard toy.

Data from an Italy cohort show wheezing episodes decreased by more than 40% among children born during the COVID-19 lockdown period versus a pre-pandemic cohort.

New international findings show BPD diagnosis in teenaged years may be a critical indicator of subsequent mental disorder into young adulthood.

Drugs like semaglutide and liraglutide were dispensed at unprecedented increased rates for adolescents and young adults over a span of just 4 years.
Approximately 1 in 5 youths who identify as a sexual gender minority will undergo a gender identity change in their adolescence; however, such changes do not appear to impact their mental health.

New cohort analyses observe a statistically raised risk of myocarditis, pericarditis and seizures among certain age groups, though the data do not go against FDA recommendations.

Abeona's pz-cel is up for indicated use to treat patients with recessive dystrophic epidermolysis bullosa.

An assessment into development scores before, during and after the COVID-19 pandemic show children aged 0 - 5 years old may only had been minimally impacted in communication and problem-solving skills.

Medicaid registry data show parents and guardians whose children have been referred to CPS are more than 3-fold likely to have a substance use disorder.
Cohort analysis shows the rate of RSV hospitalizations among children up to 5 years old skyrocketed in 2021—and then nearly doubled again in 2022—compared to pre-pandemic rates.

An intervention applied at family care practices shows benefit to tailoring education to both clinicians and medical assistants.

A novel instructional video tool was shown to effectively inform parents and guardians on fever etiology and care strategies in children being discharged from the pediatric emergency department.

The Tennessee STAT tool was associated with improved primary care provider perspective and confidence to lead, rather than refer, on pediatric autism diagnosis.

Investigators observe determinable and likely harmful levels of metabolite on children exposed to their parents' secondhand e-cigarette vapors.
A qualitative analysis shows adolescents and their parents more frequently report an unreadiness to transition from pediatric to adult type 1 diabetes care.

A cohort analysis from Japan shows that more visible symptoms—though considered less clinical by experts—may be indicative of heightened suicidal behavior in teenagers.
A new predictive model from Taiwan suggests cell-based quadrivalent flu vaccines may better egg-based options in reducing disease burden and improving the societal costs of the flu season among children and adolescents.

A new recommendation statement from the Preventive Services Task Force remains inconclusive on the benefits versus risks of asymptomatic screening for speech and language delay disorders.

The Complete Response Letter (CRL) is based on additional efficacy data required by the FDA, according to Sanofi and Regeneron.

Data suggest confirmatory trials conducted for drugs granted accelerated approval by the FDA have taken a median 3.5 years to complete since 2012.

After addressing a 2021 CRL, Verrica Pharmaceuticals vies for the agent to become the first drug approved to treat the common skin infection.

A cross-sectional study showed cannabis ingestion has been the cause of 1 in every 3 pediatric poisoning hospitalizations in provinces that sell edibles.

New surveillance data from the 2021-22 influenza show the impact of coinfection among younger patients.

An economic analysis of a clinical sleep intervention suggests it may provide a step toward improved health equity.
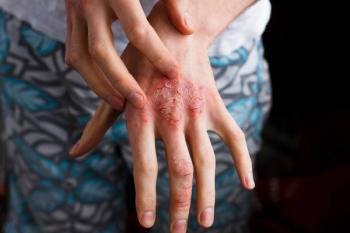
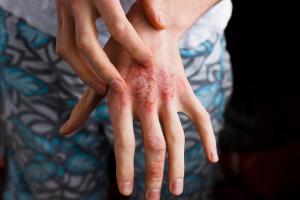
New 16-week data from the SPROUT trial show the PDE4 inhibitor may be highly efficacious in children and adolescents.
Published: October 21st 2022 | Updated:

Published: May 24th 2024 | Updated:

Published: May 24th 2024 | Updated:

Published: October 21st 2023 | Updated:

Published: February 20th 2019 | Updated:

Published: April 19th 2024 | Updated: