Atopic Dermatitis
Latest News
Latest Videos

More News

Lawrence Eichenfield, MD, reacts to the FDA's acceptance of a supplemental New Drug Application for roflumilast cream 0.15% to treat atopic dermatitis (AD) in patients aged 6 years and older.

The FDA has accepted a supplemental New Drug Application (sNDA) for roflumilast cream 0.15% for the treatment of atopic dermatitis (AD) in children aged 6 years and up.
Of patients in the atopic dermatitis (AD) cohort, 36.6% developed at least 1 comorbidity amid follow-up compared to 28.5% in the non-AD reference cohort, investigators of a Sweden, nationwide, population-based cohort study found.
Of the patients that responded to lebrikizumab at week 16 in the phase 3 trials ADvocate 1 and ADvocate 2, 84% achieved a clinically meaningful response in at least 1 domain of the disease (mild signs, symptoms, or quality of life impact) at 52 weeks.

A rapid reduction in pruritis as early as 24 hours after first application was announced as new positive data from a pair of identical, phase 3 studies of tapinarof cream 1% in children as young as 2 years and adults with atopic dermatitis (AD).
Positive topline results from a pair of identical phase 3 trials support the submission of a supplemental New Drug Application to the FDA for Arcutis Biotherapeutics’ roflumilast cream 0.15%, a once-daily topical to treat mild to moderate atopic dermatitis (AD) in children 6 years or older.
Based on recent positive phase 3 results, Arcutis Biotherapeutics,. intends to submit a supplemental New Drug Application with the FDA for roflumilast cream 0.05% to treat mild to moderate atopic dermatitis in children aged 2 to 5 years.

Peter Lio, MD, clinical assistant professor of dermatology and pediatrics at Northwestern University Feinberg School of Medicine in Chicago, Illinois, discusses the 2023 Society for Pediatric Dermatology Meeting held in Asheville, North Carolina, and what pearls to consider following the atopic dermatitis panel he took part in.

In her presentation ‘Treatment of Eczema in Pediatric Patients Under 12 Years,’ Dr. Garza-Mayers spoke on considerations for atopic dermatitis management.
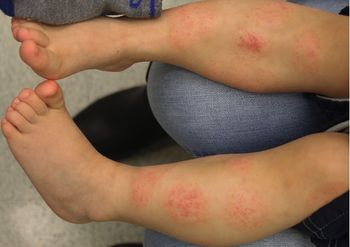
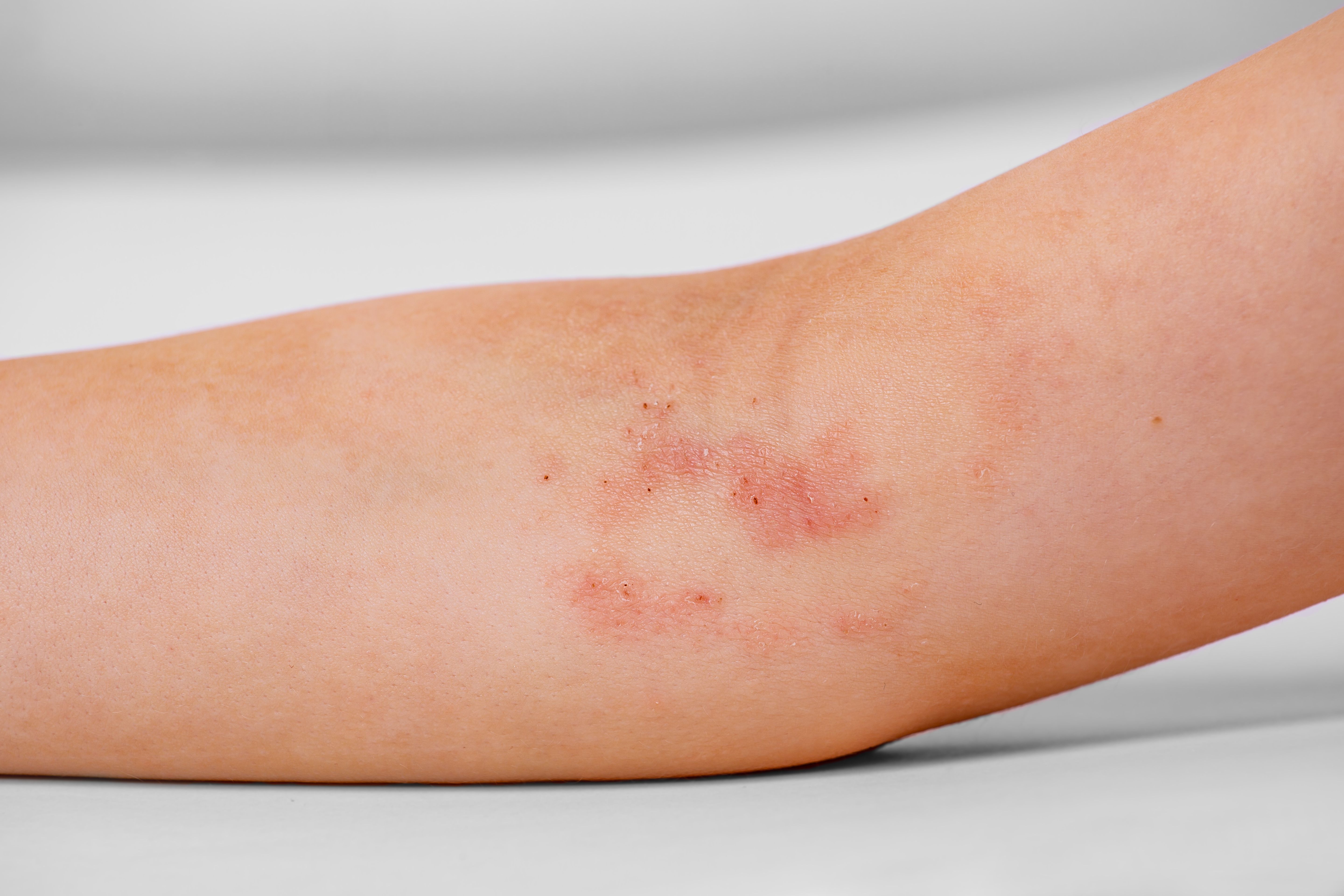
A healthy 2-year-old boy with atopic dermatitis developed intermittent flares of eczematous patches on his legs with dramatic sparing below the sock line following meals. What is the diagnosis?

The once-daily topical cream for the treatment of atopic dermatitis (AD) has demonstrated positive topline results in 2 phase 3 studies in adults and children aged 2 years and up. According to Dermavant, a supplemental new drug application (sNDA) filed with the FDA is anticipated in Q1 of 2024.
In identical phase 3 trials, roflumilast demonstrated topline positive results, reducing itch in children and adults with mild to moderate AD.
In a phase 3 clinical trial, dupilumab met both its primary and key secondary endpoints, demonstrating efficacy as a potential treatment for atopic hand and foot dermatitis.

At the 44th National Association of Pediatric Nurse Practitioners Conference, methods of treating atopic dermatitis were discussed.

A recent study indicated that lebrikizumab treatment for 16 weeks was effective for adolescents and adults with moderate-to-severe atopic dermatitis.
Results from a recent phase 3 clinical trial demonstrated topline safety and efficacy results for tapinarof cream, 1% (VTAMA; Dermavant) as a potential treatment for atopic dermatitis in patients 2 years and older.

A supplemental New Drug Application (sNDA) for abrocitinib (CIBINQO; Pfizer) in adolescents has been approved by the US Food and Drug Administration.
A recent study highlighted new information on racial disparities in health care for Black and Hispanic pediatric patients with atopic dermatitis.
Brittany Craiglow, MD, and Angela J Lamb, MD, share closing thoughts on the role of pediatricians and emerging treatments in the pipeline for pediatric AD.

Expert dermatologists review the use of dupilumab for treatment of pediatric AD, and how topical and injectable medication can impact a patient’s lifestyle.
Angela Lamb, MD, shares her approach to treatment selection for pediatric patients with AD.

Brittany Craiglow, MD, and Angela J Lamb, MD, share initial case impressions of a 7-year-old female patient with AD and discuss the key role of parents in disease management.

A recent article highlighted multiple therapies for treating atopic dermatitis, including many which do not require a prescription.

A recent article highlighted multiple forms of treatment for atopic dermatitis, detailing their benefits and adverse events.
Adults with atopic dermatitis saw improved symptoms when taking dupilumab compared to placebo, regardless of the age when they first presented with the condition.