
Drug Pipeline News
Latest News
Latest Videos

More News

FDA approves gadopiclenol extension in neonates
The gadopiclenol extension for use in infants and neonates offers high-quality MRI contrast enhancement at half the standard gadolinium dose.

Get caught up with Contemporary Pediatrics! This list helps you navigate our top stories from the week, all in one place.

Phase 3 KEPLER data showed vedolizumab achieved remission in nearly half of children with ulcerative colitis, with consistent safety and durable benefit.

FDA expands pitolisant indication to include cataplexy in patients 6 years and older with narcolepsy.

Ferric maltol gains FDA approval for children 10 years and older with iron deficiency, supported by phase 3 pediatric data.

Phase 2 data suggest FcRn blockade may reduce maternal IgG and placental antibody transfer in HDFN without excess infant infections.

Open-label data show sustained seizure reduction, stable safety, and improved quality of life in LGS treated with fenfluramine.

A study found that a nirsevimab immunization program reduced RSV-related hospitalizations by 85.9% in the first season and 55.3% in the second.

Breztri Aerosphere shows positive phase 3 data against uncontrolled asthma
Phase trials show that Breztri Aerosphere triple therapy reduces annual severe exacerbation rates vs dual-combination inhaled therapy.

Oral infigratinib linked to improved growth and proportionality in achondroplasia
In the PROPEL 3 trial, oral infigratinib significantly increased annualized height velocity and improved body proportionality in children with achondroplasia.

Phase 2b maintenance data show sustained and new EASI, vIGA-AD, and itch responses with monthly or quarterly rezpegaldesleukin.
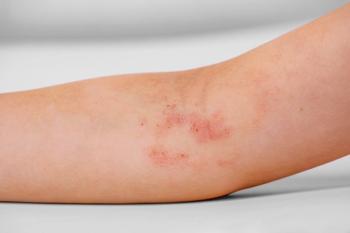
In LEVEL UP period 2, most patients who switched from dupilumab to upadacitinib achieved higher skin clearance and itch relief by 16 weeks.

The FDA declined approval of RGX-121 for MPS II, citing concerns about study design, control comparability, and surrogate end point validity.

FDA grants priority review to marstacimab for hemophilia A or B in children 6 years and older and patients with inhibitors.

A look back at the FDA submissions and regulatory decisions in the pediatric health care space from January 2026.

AbbVie submitted FDA and EMA applications for upadacitinib (RINVOQ) in adults and adolescents with non-segmental vitiligo, supported by phase 3 Viti-Up data.

Phase 2 INTEGUMENT-INFANT study results show roflumilast cream 0.05% was well tolerated and improved disease severity in infants with atopic dermatitis.

The FDA has issued a CRL for Anaphylm sublingual epinephrine, citing human factors and PK study needs; resubmission is anticipated in Q3 2026.

Get caught up with Contemporary Pediatrics! This list helps you navigate our top stories from the week, all in one place.

Three-year EMBARK data show ELEVIDYS slows functional decline in ambulatory Duchenne patients, extending earlier 2-year efficacy findings.

FDA accepted centanafadine NDA for priority review, supported by phase 3 trials showing efficacy in children, adolescents, and adults with ADHD.

Findings from phase 3 SHORE and COAST 2 trials showed amlitelimab met key efficacy end points in moderate to severe atopic dermatitis, supporting planned regulatory submissions.

FDA clears first at-home test to detect RSV, influenza, and COVID-19 in infants as young as 6 months
FDA-cleared Flowflex Plus 4-in-1 home test detects RSV, influenza A/B, and COVID-19 in adults and children as young as 6 months.

Vimal H. Prajapati, MD, discusses the latest roflumilast cream data from the INTEGUMENT-PED trial for pediatric atopic dermatitis.

New phase 3 trial data reveal fremanezumab's effectiveness and safety for preventing migraines in children, offering a vital treatment option.














