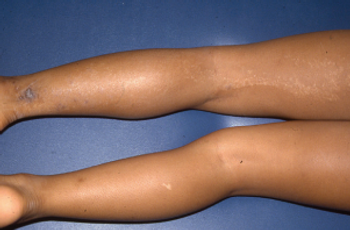
What's the diagnosis of this linear eruption on an adolescent girl's leg?

What's the diagnosis of this linear eruption on an adolescent girl's leg?

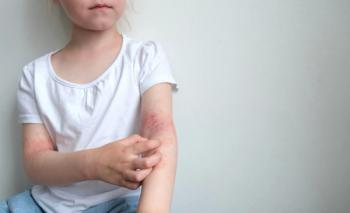
Take your best guess and try to diagnose this 9-year-old girl's linear eruption on the back of her left thigh and leg. Stay tuned for the full case presentation and correct diagnosis!
The study, known as BRAVE-AA-PEDS, was sponsored by Eli Lilly and Company and involved 257 adolescents aged 12 to under 18 years.
The newest tapinarof results were presented at the 2025 American Academy of Dermatology (AAD) Annual Meeting.
Lebrikizumab improves itch, sleep interference, and skin pain in atopic dermatitis patients previously treated with dupilumab, offering a new treatment option.

Get caught up with Contemporary Pediatrics! This list helps you navigate our top stories from the week, all in one place.

The application is for children aged 2 to 5 years with mild-to-moderate atopic dermatitis.
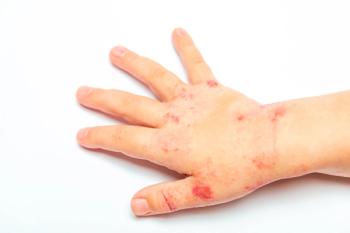
Nearly 40% of children treated with roflumilast cream 0.05% achieved a 75% improvement in EASI-75.
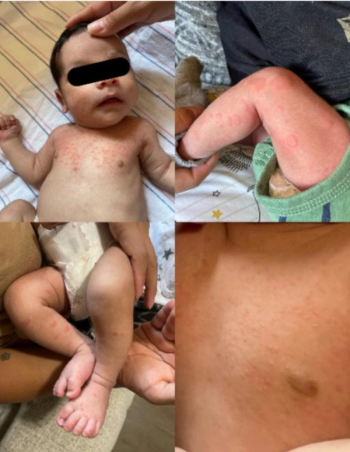
On exam, the patient is found to have circular, slightly raised, blanching, erythematous papules on the lower extremities and abdomen.

Can you diagnose this patient in our most recent puzzler poll?

Get caught up with Contemporary Pediatrics! This list helps you navigate our top stories from the week, all in one place.

Ensoma's lead program, EN-374, has been granted a pair of FDA designations for the treatment of X-linked chronic granulomatous disease.
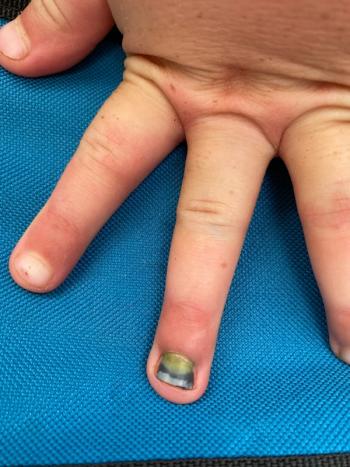
The patient's mother reported that the nail had developed a green discoloration and had begun to detach approximately 1 week prior to the visit.

Get caught up with Contemporary Pediatrics! This list helps you navigate our top stories from the week, all in one place.
The investigational biologic targets the interleukin-2 receptor complex in the body in order to stimulate proliferation of inhibitory immune cells.

A look into nutrition's role in acne care, highlighted by Colleen Sloan, PA-C, RDN.
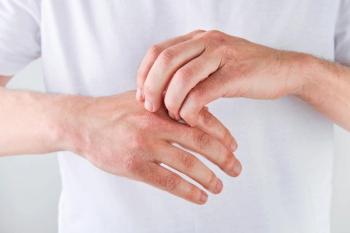
The primary and all key secondary endpoints were met compared to cream vehicle in the phase 3 DELTA TEEN trial.

Get caught up with Contemporary Pediatrics! This list helps you navigate our top stories from the week, all in one place.

The past year saw a number of approvals adding to the toolbox for atopic dermatitis, including new formulations that work in new mechanisms.

Editor-in-chief Tina Tan, MD, FAAP, FIDSA, FPIDS, highlights the January/February, 2025, issue of Contemporary Pediatrics.
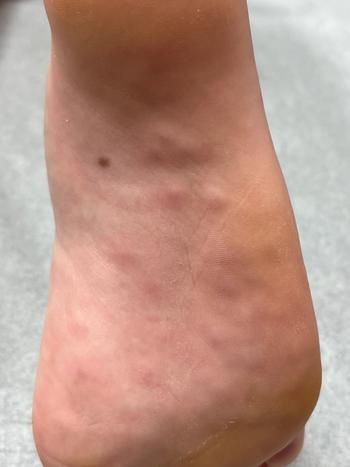
Medical advice was sought on the fifth day of symptoms, and by this time, the child was improving clinically.
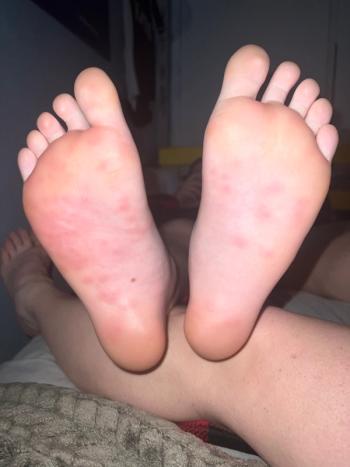
The photos in this case were taken soon after the painful and red papules appeared.

Russell Libby, MD, FAAP highlights some key benefits of roflumilast 0.15%, approved in 2024 to treat AD in patients aged 6 years and older.

Get caught up with Contemporary Pediatrics! This list helps you navigate our top stories from the week, all in one place.

"Tapinarof comes in with that mixture of the short-term studies and longer-term studies intermittently, giving us a nice, effective alternative non-steroid for eczema across the ages."