Dermatology
Latest News
Latest Videos

Shorts






Podcasts
More News
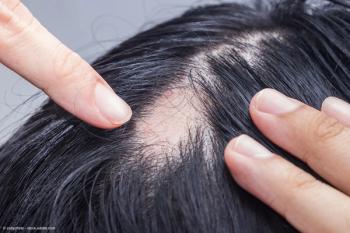
AbbVie filed an sNDA for upadacitinib to treat severe alopecia areata in adults and adolescents based on phase 3 UP-AA trial results.

FDA clearance is being sought for roflumilast cream for infants 3 months and older with mild to moderate atopic dermatitis.

MMR coverage remained below herd immunity 20 months after an Ohio outbreak, with minimal gains and persistent disparities in children.

The FDA approved dupilumab for children aged 2 to 11 years with chronic spontaneous urticaria, making it the first biologic for young pediatric CSU patients.
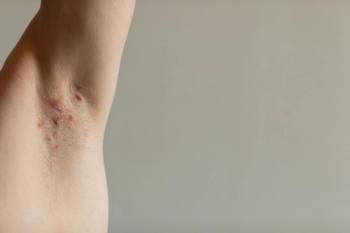
Real-world data show pediatric hidradenitis suppurativa treatment relies on antibiotics, with delayed escalation and demographic differences in timing.

A pilot study finds diverse arthritis patterns and high metabolic comorbidity burden in idradenitis suppurativa, with IL-17 inhibitors showing improved outcomes.
A review suggests that early use of topical calcineurin inhibitors, corticosteroids, and phototherapy can achieve moderate repigmentation in segmental vitiligo.

A review of 6 novel drugs approved by the FDA, with links to coverage from Contemporary Pediatrics and other MJH Life Sciences brands.

A review of 6 drugs approved by the FDA in Q1 2026, with links to coverage from Contemporary Pediatrics.

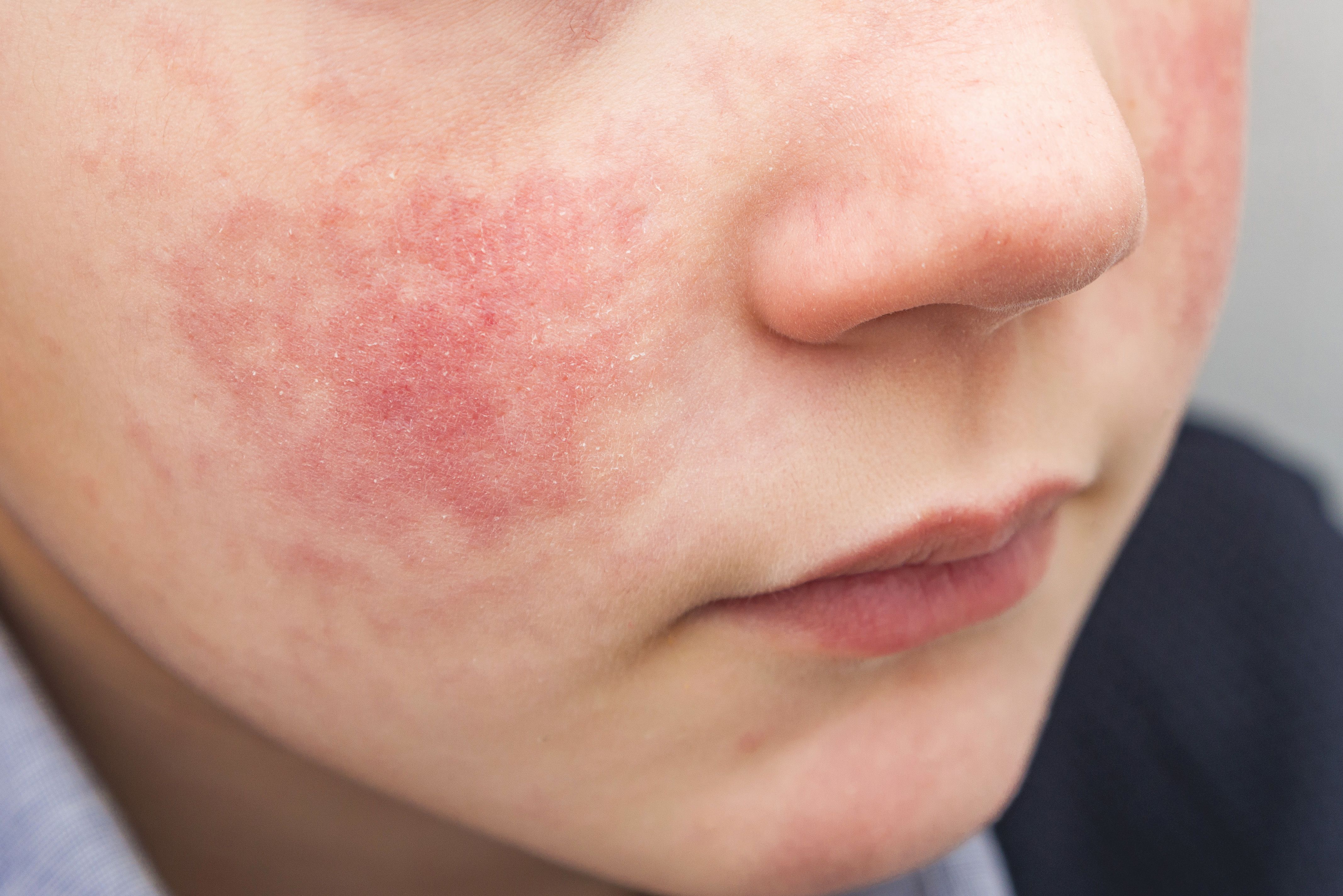
Can you correctly guess the diagnosis?

A phase 3 trial shows that lebrikizumab improves skin clearance and itch in infants and children with moderate to severe atopic dermatitis.

Secukinumab now treats moderate to severe hidradenitis suppurativa in patients 12 years and older, making it the first IL‑17A biologic option for teens.

Phase 2b maintenance data show sustained and new EASI, vIGA-AD, and itch responses with monthly or quarterly rezpegaldesleukin.
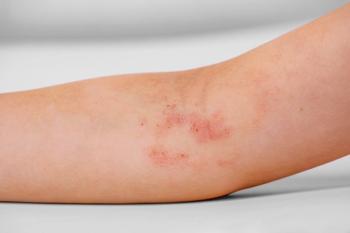
In LEVEL UP period 2, most patients who switched from dupilumab to upadacitinib achieved higher skin clearance and itch relief by 16 weeks.

AbbVie submitted FDA and EMA applications for upadacitinib (RINVOQ) in adults and adolescents with non-segmental vitiligo, supported by phase 3 Viti-Up data.

Phase 2 INTEGUMENT-INFANT study results show roflumilast cream 0.05% was well tolerated and improved disease severity in infants with atopic dermatitis.

Findings from phase 3 SHORE and COAST 2 trials showed amlitelimab met key efficacy end points in moderate to severe atopic dermatitis, supporting planned regulatory submissions.

A national survey found most patients with chronic inflammatory skin disease rely on flare-based care and report concerns about long-term topical steroid use.

Vimal H. Prajapati, MD, discusses the latest roflumilast cream data from the INTEGUMENT-PED trial for pediatric atopic dermatitis.

Join us for a recap of some of our top dermatology Puzzler cases from 2025.

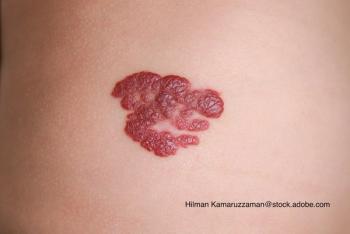
See the full case of our most recent Puzzler quiz and view the correct diagnosis of this 19-year-old woman with painful, purpuric nodules in the phalanges of her fingers.

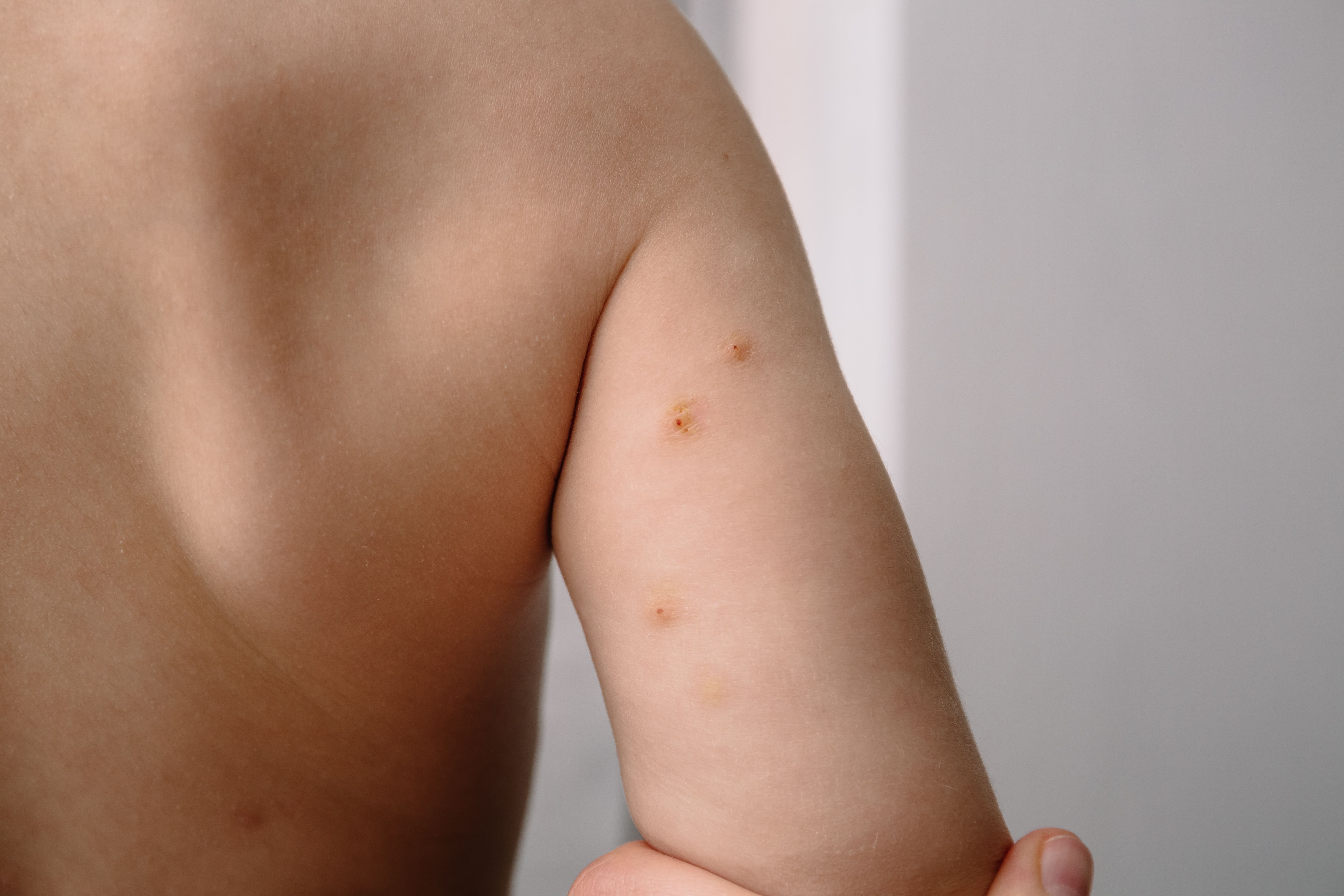
Can you guess the diagnosis?

The target action date for roflumilast cream 0.3% for plaque psoriasis in children 2 to 5 years is set for June 29, 2026.
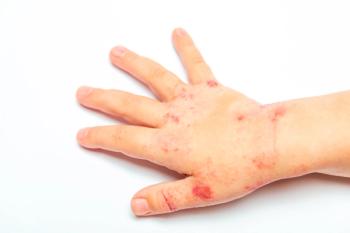
Sub-analysis of ADORING trials shows VTAMA cream improved skin clearance, symptoms, and patient-reported outcomes in children aged 2-17.
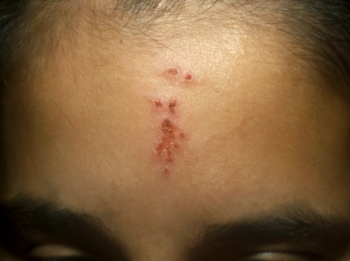
Can you guess the diagnosis?